Trimastix
Alastair Simpson and Vladimir Hampl


This tree diagram shows the relationships between several groups of organisms.
The root of the current tree connects the organisms featured in this tree to their containing group and the rest of the Tree of Life. The basal branching point in the tree represents the ancestor of the other groups in the tree. This ancestor diversified over time into several descendent subgroups, which are represented as internal nodes and terminal taxa to the right.

You can click on the root to travel down the Tree of Life all the way to the root of all Life, and you can click on the names of descendent subgroups to travel up the Tree of Life all the way to individual species.
For more information on ToL tree formatting, please see Interpreting the Tree or Classification. To learn more about phylogenetic trees, please visit our Phylogenetic Biology pages.
close boxIntroduction
Trimastix is a group of free-living heterotrophic flagellates. There are only a tiny handful of species, and their ecological role is essentially unstudied. Trimastix however is in an interesting evolutionary position as the sister group to oxymonads, a moderately diverse and well-known group of animal commensals/symbionts (see Dacks et al., 2001; Hampl et al., 2009). Like oxymonads they are anaerobes/microaerophiles that lack classical mitochondria, and much of the current interest in Trimastix concerns the evolution of mitochondrial organelles in anaerobic eukaryotes (see Hampl et al., 2008).Characteristics
General appearance, and apomorphies
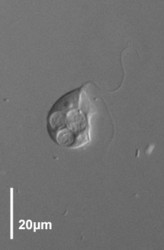
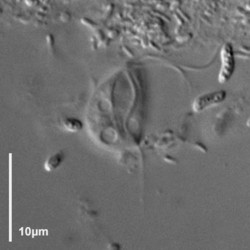
Trimastix cells are roughly pyriform (teardrop-shaped), and about 10-25 micrometres long, depending on the species (Kent, 1880-2, Klebs, 1893, Bernard et al., 2000; See title illustrations and Fig. 1). Cells are usually observed swimming freely, not attached to a surface. The flagella insert near the anterior end of the cell. Despite the name, Trimastix cells have four flagella, not three. One flagellum is directed posteriorly, another anteriorly (more or less), and two others are directed laterally – one left and one right (Last title illustration, and Fig. 1b). As a consequence it is difficult to see all four flagella at the same time, and when the genus was first described by Kent (1880-2) one flagellum was overlooked.

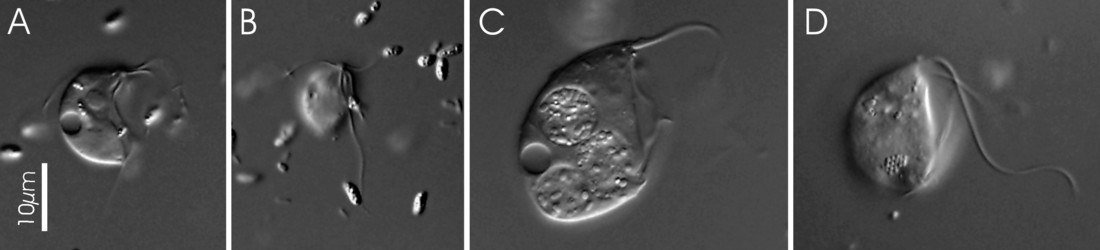
Figure 1. Light microscopy (DIC) images of Trimastix pyriformis (A,B) and Trimastix marina (C,D). ©
The posterior flagellum bears two very broad vanes with thickened margins, which can be seen most clearly by electron microscopy (Fig. 2, 3b), but can also sometimes be distinguished by light microscopy as well (Brugerolle and Patterson, 1997; O’Kelly et al., 1999; Simpson et al., 2000). In one species, Trimastix marina, the anterior flagellum is thickened as well (Simpson et al., 2000; see Fig. 1c, 2, 3a).

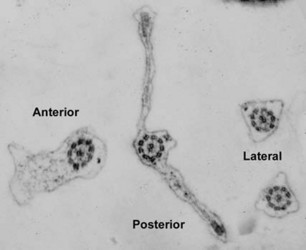
Figure 2. Trimastix marina. Transmission electron micrograph showing transverse section of all four flagella from one cell. Note the broad vanes on the posterior flagellum, with thickened margins, and the thickening of the anterior flagellum © Alastair Simpson


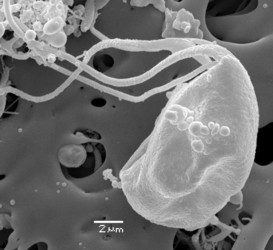
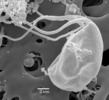
Figure 3. Trimastix marina, Scanning Electron Micrographs. A. Whole cell portrait, highlighting the ventral feeding groove. The very posterior end of the groove, deflected laterally is the opening of the cytostome - the site of ingestion. B. Mid-portion of the groove and the broad vanes on the posterior flagellum . ©
- A very broad fibrous crescent connecting dorsally the proximal ends of the left and right microtubular roots
- Substantially thickened margins to the vanes on the posterior flagellum.
- The microtubules of the inner portion of the right root are always separated rather than formed into a tight ribbon
- The C fibre (connected to the left microtubular root) includes long spoke-like structures.
Mitochondrial organelles
In place of classical mitochondria Trimastix spp. have organelles that are bounded by two membranes, but that lack mitochondrial cristae (Brugerolle and Patterson, 1997; O’Kelly et al., 1999; Simpson et al., 2000). The organelles of Trimastix also tend to be smaller than typical mitochondria, especially in Trimastix marina (Brugerolle and Patterson, 1997; O’Kelly et al., 1999; Simpson et al., 2000). Nuclear transcripts from Trimastix pyriformis include a dozen ‘mitochondrial proteins’ that are normally involved in protein import and folding, or metabolic functions (Hampl et al., 2008). This indicates that these organelles share an evolutionary history with typical mitochondria (Hampl et al., 2008). These mitochondrial proteins include all the major proteins of the glycine cleavage system, a mitochondrial pathway which is not present in toto in the mitochondrion-like organelles of well studied anaerobic eukaryotes such as Giardia, Trichomonas and Entamoeba, It is suspected that the mitochondrion-like organelles of Trimastix might function as hydrogenosomes (as in parabasalids), but this has not been demonstrated – although Trimastix does produce transcripts for signature hydrogenosomal enzymes (hydrogenase, pyruvate:ferredoxin oxidoreductase), it is not known whether these enzymes are active in the mitochondrial organelle (Hampl et al., 2008).
Discussion of Phylogenetic Relationships
There are only three recognized species of Trimastix – Trimastix marina, Trimastix pyriformis, and Trimastix inequalis. Trimastix convexa Grasse is regarded as a junior synonym of Trimastix pyriformis Klebs (see Bernard et al., 2000). Molecular phylogenies consistently group Trimastix marina and Trimastix pyriformis as a clade (Dacks et al., 2001; Simpson et al., 2006), supporting the monophyly of Trimastix, but there are no molecular data for Trimastix inequalis with which to test the internal phylogenetic relationships.
References
Bernard, C., Simpson, A.G.B. and Patterson, D.J. (2000) Some free-living flagellates (Protista) from anoxic habitats. Ophelia 52: 113-142.
Brugerolle, G and Patterson, D.J. (1997) Ultrastructure of Trimastix convexa Hollande, an amitochondriate anaerobic flagellate with a previously undescribed organization. Europ. J. Protistol. 33: 121-130.
Dacks, J.B., Silberman, J.D., Simpson, A.G.B., Moriya, S., Kudo, T., Ohkuma, M. and Redfield, R.J. (2001) Oxymonads are closely related to the excavate taxon Trimastix. Mol Biol Evol, 18, 1034-1044.
Hampl, V., Hug, L., Leigh, J.W., Dacks, J.B., Lang, B.F., Simpson, A.G.B. and Roger, A.J. (2009) Phylogenomic analyses support the monophyly of Excavata and resolve relationships among eukaryotic "supergroups". Proc Natl Acad Sci U S A, 106, 3859-3864.
Hampl, V., Silberman, J.D., Stechmann, A., Diaz-Trivino, S., Johnson, P.J. and Roger, A.J. (2008) Genetic evidence for a mitochondriate ancestry in the 'amitochondriate' flagellate Trimastix pyriformis. PLoS One, 3, e1383.
Kent, W.S. (1880-1882). A manual of the Infusoria. (3 vols) London.
Klebs, G., (1893) Über die Organisation einiger Flagellatengruppen und ihre Beziehungen zu anderen Infusorien. - Unters. Bot. Inst. Tübingen. 1: 233-262.
O'Kelly C.J., Farmer M.A. and Nerad T.A. (1999) Ultrastructure of Trimastix pyriformis (Klebs) and similarities of Trimastix species with retortamonads and jakobids. Protist 150, 149-162.
Simpson, A.G.B., Bernard, C. and Patterson, D.J. (2000) The ultrastructure of Trimastix marina Kent, 1880 (Eukaryota), an excavate flagellate. Europ. J. Protistol. 36, 229-252.
Simpson, A.G.B., Inagaki, Y. and Roger, A.J., (2006) Comprehensive multigene phylogenies of excavate protists reveal the evolutionary positions of primitive eukaryotes. Mol. Biol. Evol. 23: 615-625.
Title Illustrations

| Scientific Name | Trimastix marina |
|---|---|
| Comments | Monoprotistan culture, strain WL |
| Specimen Condition | Live Specimen |
| Identified By | Alastair Simpson |
| Image Use |
 This media file is licensed under the Creative Commons Attribution-NonCommercial License - Version 3.0. This media file is licensed under the Creative Commons Attribution-NonCommercial License - Version 3.0.
|
| Copyright |
© 2009 Alastair Simpson

|
| Scientific Name | Trimastix marina |
|---|---|
| Comments | Scanning electron micrograph of Trimastix marina trophozoite |
| Specimen Condition | Dead Specimen |
| Life Cycle Stage | Trophozoite |
| Copyright |
©

|
| Scientific Name | Trimastix pyriformis |
|---|---|
| Comments | From monoprotiston culture, strain RCP / RCP-MX |
| Specimen Condition | Live Specimen |
| Identified By | Alastair Simpson |
| Image Use |
 This media file is licensed under the Creative Commons Attribution-NonCommercial License - Version 3.0. This media file is licensed under the Creative Commons Attribution-NonCommercial License - Version 3.0.
|
| Copyright |
© 2009 Alastair Simpson

|
| Scientific Name | Trimastix pyriformis |
|---|---|
| Comments | From monoprotistan culture, strain RCP / RCP-MX |
| Specimen Condition | Live Specimen |
| Identified By | Alastair Simpson |
| Image Use |
 This media file is licensed under the Creative Commons Attribution-NonCommercial License - Version 3.0. This media file is licensed under the Creative Commons Attribution-NonCommercial License - Version 3.0.
|
| Copyright |
© 2009 Alastair Simpson

|
About This Page
This page is being developed as part of the Tree of Life Web Project Protist Diversity Workshop, co-sponsored by the Canadian Institute for Advanced Research (CIFAR) program in Integrated Microbial Biodiversity and the Tula Foundation.
Alastair Simpson

Dalhousie University, Halifax, Nova Scotia, Canada

Dalhousie University, Halifax, Nova Scotia, Canada
Correspondence regarding this page should be directed to Alastair Simpson at and Vladimir Hampl at
Page copyright © 2009 Alastair Simpson and
 Page: Tree of Life
Trimastix .
Authored by
Alastair Simpson and Vladimir Hampl.
The TEXT of this page is licensed under the
Creative Commons Attribution-NonCommercial License - Version 3.0. Note that images and other media
featured on this page are each governed by their own license, and they may or may not be available
for reuse. Click on an image or a media link to access the media data window, which provides the
relevant licensing information. For the general terms and conditions of ToL material reuse and
redistribution, please see the Tree of Life Copyright
Policies.
Page: Tree of Life
Trimastix .
Authored by
Alastair Simpson and Vladimir Hampl.
The TEXT of this page is licensed under the
Creative Commons Attribution-NonCommercial License - Version 3.0. Note that images and other media
featured on this page are each governed by their own license, and they may or may not be available
for reuse. Click on an image or a media link to access the media data window, which provides the
relevant licensing information. For the general terms and conditions of ToL material reuse and
redistribution, please see the Tree of Life Copyright
Policies.
- First online 16 November 2009
- Content changed 16 November 2009
Citing this page:
Simpson, Alastair and Vladimir Hampl. 2009. Trimastix . Version 16 November 2009. http://tolweb.org/Trimastix/97437/2009.11.16 in The Tree of Life Web Project, http://tolweb.org/









 Go to quick links
Go to quick search
Go to navigation for this section of the ToL site
Go to detailed links for the ToL site
Go to quick links
Go to quick search
Go to navigation for this section of the ToL site
Go to detailed links for the ToL site