Agaricomycotina
Jelly Fungi, Yeasts, and Mushrooms
David S. Hibbett


This tree diagram shows the relationships between several groups of organisms.
The root of the current tree connects the organisms featured in this tree to their containing group and the rest of the Tree of Life. The basal branching point in the tree represents the ancestor of the other groups in the tree. This ancestor diversified over time into several descendent subgroups, which are represented as internal nodes and terminal taxa to the right.

You can click on the root to travel down the Tree of Life all the way to the root of all Life, and you can click on the names of descendent subgroups to travel up the Tree of Life all the way to individual species.
For more information on ToL tree formatting, please see Interpreting the Tree or Classification. To learn more about phylogenetic trees, please visit our Phylogenetic Biology pages.
close boxIntroduction
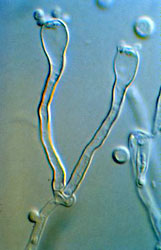
The Agaricomycotina is one of three major clades of Basidiomycota (the others are the Pucciniomycotina and Ustilaginomycotina). The Agaricomycotina contains roughly 20,000 described species, which is almost 70% of the (known) Basidiomycota. About 98% of the species of the Agaricomycotina are in a clade called the Agaricomycetes, which includes mushrooms, bracket fungi, puffballs, and others. The other major groups are the Tremellomycetes and Dacrymycetes. These latter groups include "jelly fungi" (Fig. 1), which have gelatinous, often translucent fruiting bodies (e.g., "witches butter" Tremella mesenterica), as well as many yeast-forming species (Hibbett 2007).


Figure 1. Phlogiotis (Tremiscus) helvelloides (Auriculariales).
© David S. Hibbett.
Members of the Agaricomycotina display the full range of ecological strategies that characterizes the Basidiomycota as a whole. To obtain carbon nutrition, Agaricomycotina decompose dead organic matter or enter into diverse associations (both antagonistic and benign) with plants, animals, and other fungi. Mycorrhizal associations with plants are present in many lineages of Agaricomycetes, including certain jelly fungi (Selosse et al. 2002). Mycoparasitism (parasitism of a fungus by a fungus) is especially widespread in the Tremellomycetes (Bandoni 1984). Few Agaricomycotina are medically important. An exception is Filobasidiella neoformans (also called Cryptococcus neoformans), which is a serious pathogen of immunocompromised individuals (Mitchell and Perfect 1995).
Agaricomycotina include the vast majority of edible mushrooms, most of which are in the Agaricomycetes. Several jelly fungi are cultivated for food in Asia, including the "wood ear" fungus, Auricularia auricula-judae (Fig. 2), and the white jelly fungus, Tremella fuciformis.


Figure 2. Cultivated fruiting bodies of Auricularia auricula-judae (Auriculariales).
© David S. Hibbett.
Characteristics
Agaricomycotina are quite variable in both macroscopic and microscopic features. Traditionally, one of the most important characters in the higher-level taxonomy of Basidiomycota has been the form and septation of the basidia (the cells in which meiosis occurs, and on which sexual spores are formed) (Talbot, 1968; McNabb, 1973). Basidia of Agaricomycotina have various shapes and may be undivided or divided by transverse or longitudinal septa (Fig. 3). Previously--including in a former version of this Tree of Life page--the Agaricomycotina with "homobasidia" (which are undivided basidia that produce spores on short apical appendages, called sterigmata) were classified as the "Homobasidiomycetes". However, recent phylogenetic studies suggest that clades with undivided homobasidia are interdigitated with clades that have septate "heterobasidia". Consequently, the terms "homobasidiomycetes" and "heterobasidiomycetes" are no longer used to refer to formal taxa, but rather are used as descriptive terms only (Hibbett 2007).
Most Agaricomycotina produce four spores on each basidium, but some species produce as few as one or as many as eight spores per basidium. Another important character in prior taxonomy of Basidiomycota is the presence or absence of "spore repetition", which is the production of secondary spores directly from basidiospores. The Agaricomycotina display variation in this character also. It is therefore not surprising that the Agaricomycotina, as delimited here, was not formally recognized as a taxonomic group before the advent of molecular characters.

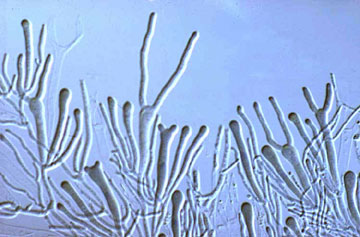
Figure 3. Two-spored "tuning fork" basidia of the jelly fungus Dacrymyces.
© George Barron. From George Barron's Website on Fungi used with permission.
One unifying morphological feature of the Agaricomycotina is the presence of "dolipore" septa, which are pores in the crosswalls between adjacent cells that are bordered by a collar-like margin of cell wall material (Fig. 4). Other Basidiomycota also have septal pores, but their structure is not the same. Flanking the dolipores in almost all Agaricomycotina is a membrane-bound structure called a "septal pore cap". These structures are also called "parenthesomes", because they resemble parentheses around the septal pores when viewed in thin sections with transmission electron microscopy.
Variation in the form of the parenthesomes has provided clues to phylogenetic relationships in the Agaricomycotina (Adams et al. 1995; Bandoni and Oberwinkler 1982; Hibbett and Thorn 2001; McLaughlin et al. 1995; Moore, 1985; Swann and Taylor 1993; Wells and Bandoni 2001). Tremellomycetes have "vesiculate" parenthesomes, in which the septal pore cap is divided into cup-shaped sections. Dacrymycetes and some Agaricomycetes have "imperforate" parenthesomes, in which the septal pore cap forms a smooth, dome-shaped covering over the dolipore. Most Agaricomycetes have "perforate" parenthesomes, in which the septal pore cap has openings of varying sizes, and appears discontinuous in section.

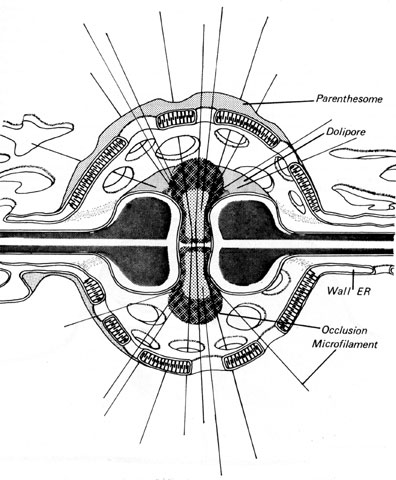
Figure 4. Dolipore septa with perforate parenthesome (septal pore cap).
From Moore and Marchant (1972); used with permission.
Most known species of Agaricomycotina are filamentous and produce a multicellular fruiting body. The fruiting body is not always what most people would regard as a "mushroom", however. For example, in "resupinate" species the fruiting body is a flattened structure, which may be little more than a thin layer of hyphae produced on the underside of a log or some other inconspicuous location. As noted, some Agaricomycotina are yeasts and produce no fruiting body at all. Fruiting bodies of Agaricomycotina reach their greatest size, diversity, and complexity in the Agaricomycetes. It is likely that the yeasts and resupinate forms have been undercollected, relative to their more conspicuous relatives.
Discussion of Phylogenetic Relationships
As in all groups of Fungi, understanding of phylogenetic relationships in Agaricomycotina has advanced rapidly in recent years. Early phylogenetic analyses using rRNA gene sequences resolved many major groups within the Hymenomycetes, but higher-level relationships among these groups were often not resolved with confidence (Fell, 2001; Swann and Taylor, 1993; Swann et al. 1995). Recently, multi-locus analyses combining rRNA and protein-coding genes have improved resolution of the deepest splits in the Agaricomycotina (Matheny et al. 2006, 2007, Hibbett et al. 2007).
The phylogenetic hypothesis and classification adopted here are based on multiple independent analyses, summarized by Hibbett et al. (2007). Three major clades are formally named: Tremellomycetes, Dacrymycetes, and Agaricomycetes (which is here taken to include the heterobasidiomycetous Auriculariales and Sebacinales). Several analyses, including a multi-locus study by Matheny et al. (2007), suggest that the Dacrymycetes and Agaricomycetes form a monophyletic group, which Swann and Taylor (1995) called the Hymenomycetidae (here, the clade is unnamed). Earlier studies using rRNA genes did not support that grouping, however (Begerow et al. 1997; Weiß and Oberwinkler 2001).
To illustrate some of the changes that have taken place in phylogenetic reconstruction and classification in Agaricomycotina (and to help you make sense of older literature), the previous classifications of Agaricomycotina in the Tree of Life and the Dictionary of the Fungi 9th edition (Kirk et al. 2001) are presented below. In the former ToL classification, the Agaricomycotina was called the Hymenomycetes, following Swann and Taylor (1993), whereas in the Dictionary of the Fungi 9th ed. the group was called the Basidiomycetes (not to be confused with "basidiomycetes", which is a general term for all Basidiomycota). The Tremellomycetidae and Dacrymycetales of the previous ToL classification are equivalent to the Tremellomycetes and Dacrymycetes (respectively) of the current ToL classification. The current Agaricomycetes includes the Homobasidiomycetes and Auriculariales of the former ToL classification. The Tremellomycetidae of the Dictionary of the Fungi 9th edition is a paraphyletic group composed of "heterobasidiomycetes" (see Characteristics, above).
Table 1. Comparison of the previous classification of Agaricomycotina in the Tree of Life and that of Kirk et al. (2001).
| Previous ToL classification | Kirk et al. (2001) |
|---|---|
|
|
The most ancient divergences in the Agaricomycotina have been particularly difficult to resolve using only rRNA genes. For example, the tree below shows results of a study by Weiß and Oberwinkler (2001), who analyzed partial nuclear large subunit (nuc-lsu) rRNA gene sequences (names in the tree are based on the previous ToL classification). One source of error in earlier rRNA-based studies is an elevated rate of evolution of nuclear rRNA genes in some members of the Cantharellales, which is an early-diverging clade in the Agaricomycetes. Specifically, members of Cantharellus and Craterellus (which include edible chanterelles) and Tulasnella (a group of resupinate "crust fungi") have highly divergent nuc rRNA sequences that cannot be aligned in their entirety to other Agaricomycetes and produce conspicuously long branches in phylograms (Hibbett et al. 1997, Matheny et al. 2007, Moncalvo et al. 2007). Analyses using protein-coding genes leave little doubt that these taxa are in the Cantharellales clade of the Agaricomycetes (Matheny et al. 2007, Moncalvo et al. 2007).


Figure 5. Schematic representation of the phylogenetic tree of Weiß and Oberwinkler (2001). Numbers along branches indicate bootstrap frequencies. Bootstrap values below 60% are not shown. Taxon names are based on the previous classification in the Tree of Life.
References
Adams, G. C., Klomparens, K. L., Hennon, P. E. 1995. Unusual reticulated parenthesomes surround the dolipore of a hyphomycete with clamp connections, Ditangifibulae dikaryotae gen. et sp. nov. Mycologia 87:909-921.
Alexopoulos, C.J., Mims, C.W. and Blackwell, M. 1996. Introductory Mycology. John Wiley and Sons, New York.
Bandoni, R. J. 1984. The Tremellales and Auriculariales: and alternative classification. Trans. Mycol. Soc. Japan 25:489-530.
Bauer, R., Begerow, D., Oberwinkler, F., Piepenbring, M. and Berbee, M. L 2001. Ustilaginomycetes. Pp. 57-84. In: The Mycota VII. Systematics and Evolution. Part B. (Mclaughlin, D. J., McLaughlin, E. G. and Lemke, P. A., eds.). Springer-Verlag, Berlin.
Begerow, D., Bauer, R., and Oberwinkler, F. 1997. Phylogenetic studies on nuclear large subunit ribosomal DNA sequences of smut fungi and related taxa. Can. J. Bot. 75:2045-2056.
Bruns, T. D., Szaro, T. M., Gardes, M., Cullings, K.W., Pan, J. J., Taylor, D. L., Horton, T. R., Kretzer, A., Garbelotto, M., Li, Y. 1998. A sequence database for the identification of ectomycorrhizal basidiomycetes by phylogenetic analysis. Molec Ecol 7:257-272.
Chen, C.-J. 1998. Morphological and molecular studies in the genus Tremella. Bibliotheca Mycologica 174:1-225.
Fell, J. W., Boekhout, T., Fonseca, A. and Sampaio J.P. 2001. Basidiomycetous yeasts. Pp. 1-36. In: The Mycota VII. Systematics and Evolution. Part B. (Mclaughlin, D. J., McLaughlin, E. G. and Lemke, P. A., eds.). Springer-Verlag, Berlin.
Hibbett, D. S. 2006. A Phylogenetic overview of the Agaricomycotina. Mycologia 98: 917-925.
Hibbett, D. S. and Binder, M. 2002. Evolution of complex fruiting-body morphologies in homobasidiomycetes. Proc. R. Soc. Lond. B 269:1963-1969.
Hibbett, D. S., M. Binder, J. F. Bischoff, M. Blackwell, P. F. Cannon, O. E. Eriksson, S. Huhndorf, T. James, P. M. Kirk, R. Lücking, T. Lumbsch, F. Lutzoni, P. B. Matheny, D. J. Mclaughlin, M. J. Powell, S. Redhead, C. L. Schoch, J. W. Spatafora, J. A. Stalpers, R. Vilgalys, M. C. Aime, A. Aptroot, R. Bauer, D. Begerow, G. L. Benny, L. A. Castlebury, P. W. Crous, Y.-C. Dai, W. Gams, D. M. Geiser, G. W. Griffith, C. Gueidan, D. L. Hawksworth, G. Hestmark, K. Hosaka, R. A. Humber, K. Hyde, J. E. Ironside, U. Kõljalg, C. P. Kurtzman, K.-H. Larsson, R. Lichtwardt, J. Longcore, J. Miądlikowska, A. Miller, J.-M. Moncalvo, S. Mozley-Standridge, F. Oberwinkler, E. Parmasto, V. Reeb, J. D. Rogers, C. Roux, L. Ryvarden, J. P. Sampaio, A. Schüßler, J. Sugiyama, R. G. Thorn, L. Tibell, W. A. Untereiner, C. Walker, Z. Wang, A. Weir, M. Weiß, M. M. White, K. Winka, Y.-J. Yao, and N. Zhang. 2007. A higher-level phylogenetic classification of the Fungi. Mycological Research 111: 509-547.
Hibbett, D. S. and Donoghue, M. J. 2001. Analysis of character correlations among wood decay mechanism, mating systems, and substrate ranges in homobasidiomycetes. Syst. Biol. 50:215-242.
Hibbett, D. S., Pine, E. M., Langer, E., Langer, G. and Donoghue, M. J. 1997. Evolution of gilled mushrooms and puffballs inferred from ribosomal DNA sequences. Proc. Nat. Acad. Sci. USA 94:12002-12006.
Hibbett, D. S. and Thorn, R. G. 2001. Homobasidiomycetes. Pp. 121-170. In: The Mycota VII. Systematics and Evolution. Part B. (Mclaughlin, D. J., McLaughlin, E. G. and Lemke, P. A., eds.). Springer-Verlag, Berlin.
Ingold, C. T. 1985. Observations on spores and their germination in certain heterobasidiomycetes. Trans. Br. Mycol. Soc. 85:417-423.
Jülich, W. 1981. Higher taxa of basidiomycetes. J. Cramer, Leichtenstein.
Kirk, P.M., Cannon, P.F., David, J.C., and Stalpers, J. 2001. Ainsworth and Bisby’s Dictionary of the Fungi. 9th ed. CAB International, Wallingford, UK.
Langer E. 1998. Evolution of Hyphodontia (Corticiaceae, Basidiomycetes) and related Aphyllophorales inferred from ribosomal DNA sequences. Folia Cryptog. Estonica Fasc. 33:57-62.
Matheny, P. B., J. A. Gossman, P. Zalar, T. K. Arun Kumar, and D. S. Hibbett. 2006. Resolving the phylogenetic position of the Wallemiomycetes: an enigmatic major lineage of Basidiomycota. Canadian Journal of Botany 84: 1794-1805.
Matheny, P. B., Z. Wang, M. Binder, J. M. Curtis, Y. W. Lim. R. H. Nilsson, K. W. Hughes, R. H. Petersen, V. Hofstetter, J. F. Ammirati, C. Schoch, G. E. Langer, D. J. McLaughlin, A. W. Wilson, P. E. Crane, T. Frøslev, Z. W. Ge, R. W. Kerrigan, J. C. Slot, E. C. Vellinga, Z. L. Liang, M. C. Aime, T. J. Baroni, M. Fischer, K. Hosaka, K. Matsuura, M. T. Seidl, J. Vaura, and D. S. Hibbett. 2007. Contributions of rpb2 and tef1 to the phylogeny of mushrooms and allies (Basidiomycota, Fungi). Molecular Phylogenetics and Evolution in press.
McLaughlin, D.J., Frieders, E.M. and Lü, Haisheng. 1995. A microscopist's view of heterobasidiomycete phylogeny. Stud. Mycol. 38: 91-109.
McNabb, R. F. R. 1973. Phragmobasidiomycetidae: Tremellales, Auriculariales, Septobasidiales. Pp. 303-316 in G. C. Ainsworth, F. K. Sparrow and A. S. Sussman (eds.) The Fungi. An advanced Treatise. Vol IVB. Academic press, New York.
McNabb, R. F. R., and Talbot, P. H. B. 1973. Holobasidiomycetidae: Exobasidiales, Brachybasidiales, Dacrymycetales, Tulasnellales. Pp. 317-326 in G. C. Ainsworth, F. K. Sparrow and A. S. Sussman (eds.) The Fungi. An advanced Treatise. Vol IVB. Academic press, New York.
Mitchell T. G. and Perfect, J. R. 1995. Cryptococcosis in the era of AIDS – 100 years after the discovery of Cryptococcus neoformans. J. Clin. Microbiol. Rev. 8:515-548.
Moncalvo J-M, Nilsson RH, Koster B, Dunham SM, Bernauer T, Matheny PB, McLenon T, Margaritescu S, Weiß M, Garnica S, Danell E, Langer G, Langer E, Larsson E, Larsson K-H, and Vilgalys R. 2007. The cantharelloid clade: dealing with incongruent gene trees and phylogenetic reconstruction methods. Mycologia 98: 937948.
Moore, R. T. 1985. The challenge of the dolipore/parenthesome septum. Pp. 175-212, in: D. Moore, L. A. Casselton, D. A. Wood, and J. Frankland (eds.) Developmental biology of higher fungi. Cambridge University Press, Cambridge.
Moore, R. T. and Marchant, R. 1972. Ultrastructural characterization of the basidiomycete septum of Polyporus biennis. Can. J. Bot. 50:2463-2469.
Müller, W. H., Stalpers, J. A., van Aelst, A. C., van der Krift, T. P., and Boekhout, T. 1998. Field emission gun-scanning electron microscopy of septal pore caps of sected species in the Rhizoctonia s.l. complex. Mycologia 90:170-179.
Oberwinkler, F. 1985 Anmerkungen zur Evolution und Systematik der basidiomyceten. Bot. Jahrb. Syst. 107, 541-580.
Oberwinkler, F. and Bandoni, R. J. 1982. A taxonomic survey of the gasteroid, auricularioid Heterobasidiomycetes. Can. J. Bot. 60:1762-1750.
Parmasto, E. 1986 On the origin of hymenomycetes (what are corticioid fungi?). Windahlia 16, 3-19.
Prillinger, H. Dörfler, C. Laaser, G. and Hauska, G. 1990. A contribution to the systematics and evolution of higher fungi: yeast-types in the basidiomycetes. Part III: Ustilago-type. Z. Mycol. 56: 251-278.
Prillinger, H., Laaser, G., Dörfler, C. and Ziegler, K. 1991. A contribution to the systematics and evolution of higher fungi: yeast-types in the basidiomycetes. Part IV: Dacrymyces-type, Tremella-type. Sydowia 43: 170-218.
Selosse, M.A., Bauer. R. and Moyersoen, B. 2002. Basal hymenomycetes belonging to Sebacinaceae are ectomycorrhizal on temperate deciduous trees. New Phytol. 155:183-195.
Smith, S. E. and Read, D. J. 1997. Mycorrhizal symbiosis. Academic Press, San Diego.
Swann, E.C. and Taylor, J.W. 1993. Higher taxa of basidiomycetes: an 18S rRNA gene perspective. Mycologia 85: 923-936.
Swann, E.C. and Taylor, J.W. 1995. Phylogenetic perspectives on basidiomycete systematics: evidence from the 18S rRNA gene. Canad. J. Bot. 73: S862-S868.
Talbot, P. H. B. 1968. Fossilized pre-Patouillardian taxonomy? Taxon 17:620-628.
Warcup, J. H. 1988. Mycorrhizal associations of isolates of Sebacina vermifera. New Phytol. 110:227-231.
Webster, J., Davey, R.A., Duller, G.A. and Ingold, C.T. 1984b. Ballistospore discharge in Itersonilia perplexans. Trans. Br. Mycol. Soc. 82: 13-29.
Weiß, M. and Oberwinkler, F. O. 2001. Phylogenetic relationships in Auriculariales and related groups-hypotheses derived from nuclear ribosomal DNA sequences. Mycol. Res. 105:403-415.
Wells, K. 1994. Jelly fungi, then and now! Mycologia 86:18-48.
Wells, K., and Bandoni, R. J. 2001. Heterobasidiomycetes. Pp. 85-120. In: The Mycota VII. Systematics and Evolution. Part B. (Mclaughlin, D. J., McLaughlin, E. G. and Lemke, P. A., eds.). Springer-Verlag, Berlin.
Information on the Internet
- Mycological Society of America. Extensive links to sites concerned with Basidiomycota and other fungi.
- The WWW Virtual Library: mycology.
- Deep Hypha Research Coordination Network. Deep Hypha is a project to coordinate and provide resources for research in fungal systematics.
- AFTOL: Assembling the Fungal Tree of Life. Collaborative research in fungal phylogenetics.
- Tom Volk's Fungi. An amazing collection of information, images, and lore about fungi.
- Australian National Botanic Gardens Fungi Web Site. Jelly fungi pages.
- Fungi of California Jelly Fungi page.
- Doctor Fungus Cryptococcus neoformans (Filobasidiella neoformans) page.
- MykoWeb. Specializing in the fungi of California.
- George Barron's Website on Fungi. Diverse fungal images by a distinguished mycologist.
- Duke University Mycology Research Unit. Information and resources about the medically important hymenomycete, Filobasidiella (Cryptococcus) neoformans.
Title Illustrations

| Scientific Name | Calocera viscosa |
|---|---|
| Comments | Dacrymycetes |
| Acknowledgements | Image used with permission |
| Source Collection | MykoWeb |
| Copyright | © Mike Wood |
| Scientific Name | Filobasidiella (Cryptococcus) neoformans |
|---|---|
| Comments | Tremellomycetes |
| Acknowledgements | Image from the Duke University Mycology Research Unit; used with permission |
| Copyright | © Wiley Schell |
| Scientific Name | Gomphus |
|---|---|
| Comments | Agaricomycetes |
| Image Use |
 This media file is licensed under the Creative Commons Attribution-NonCommercial License - Version 3.0. This media file is licensed under the Creative Commons Attribution-NonCommercial License - Version 3.0.
|
| Copyright |
© 2003 David S. Hibbett

|
About This Page
Development of this page was facilitated by the "Deep Hypha" Research Coordination Network and the "Assembling the Fungal Tree of Life" project (NSF awards DEB-0090301 and DEB-0228657). George Barron, Mike Wood, Wiley Schell, and the NRC Research Press are thanked for permission to reproduce the images in Figs. 3 and 4 and the title illustration.
David S. Hibbett

Clark University, Worcester, Massachusetts, USA
Correspondence regarding this page should be directed to David S. Hibbett at
Page copyright © 2003 David S. Hibbett
 Page: Tree of Life
Agaricomycotina. Jelly Fungi, Yeasts, and Mushrooms.
Authored by
David S. Hibbett.
The TEXT of this page is licensed under the
Creative Commons Attribution-NonCommercial License - Version 3.0. Note that images and other media
featured on this page are each governed by their own license, and they may or may not be available
for reuse. Click on an image or a media link to access the media data window, which provides the
relevant licensing information. For the general terms and conditions of ToL material reuse and
redistribution, please see the Tree of Life Copyright
Policies.
Page: Tree of Life
Agaricomycotina. Jelly Fungi, Yeasts, and Mushrooms.
Authored by
David S. Hibbett.
The TEXT of this page is licensed under the
Creative Commons Attribution-NonCommercial License - Version 3.0. Note that images and other media
featured on this page are each governed by their own license, and they may or may not be available
for reuse. Click on an image or a media link to access the media data window, which provides the
relevant licensing information. For the general terms and conditions of ToL material reuse and
redistribution, please see the Tree of Life Copyright
Policies.
- First online 22 September 2003
- Content changed 20 April 2007
Citing this page:
Hibbett, David S. 2007. Agaricomycotina. Jelly Fungi, Yeasts, and Mushrooms. Version 20 April 2007. http://tolweb.org/Agaricomycotina/20531/2007.04.20 in The Tree of Life Web Project, http://tolweb.org/











 Go to quick links
Go to quick search
Go to navigation for this section of the ToL site
Go to detailed links for the ToL site
Go to quick links
Go to quick search
Go to navigation for this section of the ToL site
Go to detailed links for the ToL site