Piroplasmorida
Jan Votýpka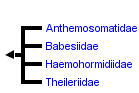


This tree diagram shows the relationships between several groups of organisms.
The root of the current tree connects the organisms featured in this tree to their containing group and the rest of the Tree of Life. The basal branching point in the tree represents the ancestor of the other groups in the tree. This ancestor diversified over time into several descendent subgroups, which are represented as internal nodes and terminal taxa to the right.

You can click on the root to travel down the Tree of Life all the way to the root of all Life, and you can click on the names of descendent subgroups to travel up the Tree of Life all the way to individual species.
For more information on ToL tree formatting, please see Interpreting the Tree or Classification. To learn more about phylogenetic trees, please visit our Phylogenetic Biology pages.
close boxIntroduction
Order Piroplasmorida Wenyon, 1926
Piroplasms are species-rich haemosporidians, owing their name to pear-shaped (pyriform) intracellular stages formed in the host erythrocytes. Some piroplasmids cause severe diseases in ruminants, horses, pigs, dogs, cats, wild mammals and, exceptionally, in humans (Babesia microti, B. divergens, and the newly described B. duncani). They were found also in birds and reptiles. These diseases lead to fevers and often kill the host. The symptoms are usually hyperthermia, anemia and hemoglobinuria. Causative agents were discovered in 1888 by Victor Babeş. Vectors of these blood-parasitizing stages are the ixodid ticks, the involvement of which was also detected rather early by Smith and Kilborne in 1893. The complete and rather complicated life cycles, however, were described in the last 30 years.
The East Coast fever and tropical theileriosis in cattle and water buffaloes are caused by Theileria parva and Theileria annulata, respectively. Several Babesia species are responsible for babesiosis of cattle, horses, dogs, and rarely also humans. Poor growth, low milk production, and mortality of infected animals resulted in several efforts to control piroplasmoses. Before implementation of successful eradication programs focused on vectors, the direct and indirect annual loss caused by the piroplasmosis was estimated at more than $100 millions in the U.S. only. While under control in developed countries, these diseases still cause serious economical loss in tropical and subtropical countries. Since the eradication of tick vectors is not realistic in most tropical countries, there is a demand for effective control of piroplasmoses by alternative approaches. Vaccines using live Theileria parasites, soluble antigen from Babesia species or vaccines composed of subunits are being developed or have even reached the stage of clinical trials, but have yet to be tested on a large scale. Recombinant forms of T. parva and T. annulata sporozoite surface antigens induce protection against parasite challenge in cattle. In the future, vaccines might be improved by inclusion of tick peptides in multivalent vaccines.
Characteristics
Members of the Piroplasmasida represent a moderately consistent group of vertebrate blood cell parasites, which can be either piriform, or pear-shaped, round, amoeboid, or rod-shaped in morphology. Unique morphological features of piroplasmids are the absence of a conoid and the reduction of the apical complex to the polar ring. Extreme reduction is characteristic for the family Theileriidae, which lacks subpellicular microtubules, the inner membrane complex, as well as the micronemes. Blood protozoans of the genera Babesia and Theileria represent worldwide distributed parasites of both domestic animals and wildlife with zoonotic potential. So far, over 30 species of piroplasms have been described from the common domestic animals.
After entering the host cell, piroplasms escape from the parasitophorous vacuole and, with few exceptions (T. buffeli and T. separata), digest host hemoglobin without producing any pigment or other visible residues. For those reasons, piroplasms are generally called non-pigment forming haemoparasites. The heteroxenous life cycle is composed of merogony taking place in a wide range of mammals (to a lesser extent in birds and reptiles), with gamogony and sporogony occurring in the gut and salivary glands of invertebrates, respectively. So far, only hard ticks (Ixodidae) were identified as vectors, although for the majority of species, vectors are yet to be found.

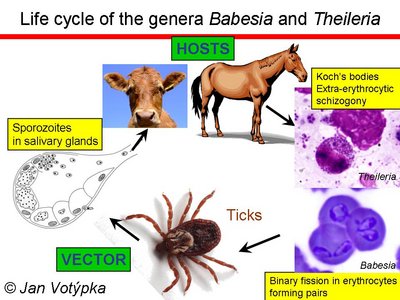
Life cycle of the genera Babesia and Theileria. © Jan Votýpka
Piroplasms are generally considered as highly specific parasites on the level of both intermediate (vertebrate) and definitive (tick serving as a vector) host. High specificity on the level of vectors has been repeatedly documented; however, the level of host specificity in their vertebrate hosts may be lower than described.
Discussion of Phylogenetic Relationships
Previous authors such as Babeş described the formation of parasites inside the red blood cells as coccal (Babesia) or bacillus-like (Theileria) bacteria. Lately it was confirmed that these pathogens are true protozoans, although their exact systematic position remained unclear for along time. Present systematics of piroplasms is based almost entirely upon criteria derived from taxonomic interpretation of a combination of morphology of the intraerythrocytic stages, host specificity, vector, mode of pathogenesis and pathogenicity, cross-immunity, frequency of schizonts and piroplasms, and spatial distribution.
The phylogenetic relationships within the order Piroplasmorida Wenyon, 1926 and the systematic position of the whole group and even the names of the genera and species of these piroplasms have been subject to constant revision and ongoing debate, and opinions have remained divergent to date. Historically, several genera had been described as belonging to the piroplasms: Theileria and Rangelia belonging to the family Theileriidae, which is characterized by preerythrocytic stages in lymphocytes, and Babesia, Nicollia, Nuttallia, Smithia, Rossiela, and Gonderia belonging to the family Babesiida and differing morphologically in the aspect of their intraerythrocytic form. Additionally, five more genera have been distinguished: Microbabesia, Babesiella, Achromaticus, Cytotauxzoon, and Piroplasma. The system has been recently reduced to the family Theileriidae du Toit 1918 comprising only the genus Theileria Bettencourt, Franca and Borges, 1907, consisting of 40 species. The family Babesiida Poche, 1913 contains the genus Babesia Starcovici, 1893 containing more than 110 species, and additionally the monotypic genera Echinozoon Garnham, 1951 and Entopolypoides Mayer, 1933. A third and forth family Anthemosomatidae Levine, 1981 (monotypic) and Haemohormidiidae Levine, 1984 with two genera have also been erected, but the life cycle and vectors of their members remain unknown.
The species belonging to the two well-accepted genera, Theileria and Babesia, seem to be significantly separated from each other by several biological features. However, the intraerythocytic stages of some species within these genera may have similar morphology. In vertebrate hosts Theileria species have a preerythrocytic development within lymphatic cells, while such schizogony is lacking in Babesia species. In ticks various stages of piroplasms enter not only the salivary glands, but also several other organs of their vectors and specifically in the genus Babesia even the eggs. Parasites are thus transmitted not only by tick saliva (alimentary infection) to vertebrate hosts but also trans-stadially (from larva to nymph) and trans-ovarially (from egg to larva); the latter path of transmission is absent in Theileria species. Molecular evidence obtained in recent studies, however, indicates that phylogenetic position and assignment of at least some species (e.g. Babesia microti, B. rodhaini, B. gibsoni and etc.) are problematic and these parasites differ from both Theileria and Babesia.The validity of the genus Cytauxzoon remains unclear. It contains the exoerythrocytic stages in its life cycle, which distinguish it from the genus Babesia. Cytauxzoon is transmitted by Dermacentor ticks. It occurs naturally in felines and causes a uniformly fatal disease in domestic cats. Two species are known, Cytauxzoon felis and C. manul. Its life cycle in the vertebrate host includes an intra-erythrocytic phase and a tissue phase consisting of large schizonts that develop in macrophages or monocytes, thus this piroplasm belongs to the family Theileriidae. However, its phylogenetic position is closely related to the above-mentioned species (e.g. Babesia gibsoni).
Information on the Internet
micro*scope: PiroplasmoridaTitle Illustrations

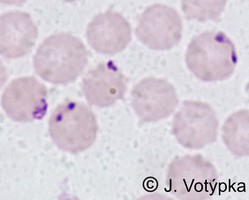
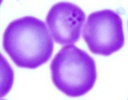
| Scientific Name | Theileria sp. from donkey |
|---|---|
| Image Use |
 This media file is licensed under the Creative Commons Attribution-NonCommercial License - Version 3.0. This media file is licensed under the Creative Commons Attribution-NonCommercial License - Version 3.0.
|
| Copyright | © 2008 Moneeb Qablan |
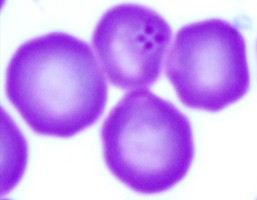
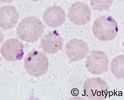
| Scientific Name | Babesia divergens |
|---|---|
| Image Use |
 This media file is licensed under the Creative Commons Attribution-NonCommercial License - Version 3.0. This media file is licensed under the Creative Commons Attribution-NonCommercial License - Version 3.0.
|
| Copyright |
© Jan Votýpka

|
About This Page
This page is being developed as part of the Tree of Life Web Project Protist Diversity Workshop, co-sponsored by the Canadian Institute for Advanced Research (CIFAR) program in Integrated Microbial Biodiversity and the Tula Foundation.
Jan Votýpka

Department of Parasitology, Charles University
Correspondence regarding this page should be directed to Jan Votýpka at
Page copyright © 2011 Jan Votýpka
 Page: Tree of Life
Piroplasmorida .
Authored by
Jan Votýpka.
The TEXT of this page is licensed under the
Creative Commons Attribution-NonCommercial License - Version 3.0. Note that images and other media
featured on this page are each governed by their own license, and they may or may not be available
for reuse. Click on an image or a media link to access the media data window, which provides the
relevant licensing information. For the general terms and conditions of ToL material reuse and
redistribution, please see the Tree of Life Copyright
Policies.
Page: Tree of Life
Piroplasmorida .
Authored by
Jan Votýpka.
The TEXT of this page is licensed under the
Creative Commons Attribution-NonCommercial License - Version 3.0. Note that images and other media
featured on this page are each governed by their own license, and they may or may not be available
for reuse. Click on an image or a media link to access the media data window, which provides the
relevant licensing information. For the general terms and conditions of ToL material reuse and
redistribution, please see the Tree of Life Copyright
Policies.
- First online 10 July 2006
- Content changed 18 May 2011
Citing this page:
Votýpka, Jan. 2011. Piroplasmorida . Version 18 May 2011 (under construction). http://tolweb.org/Piroplasmorida/68060/2011.05.18 in The Tree of Life Web Project, http://tolweb.org/





 Go to quick links
Go to quick search
Go to navigation for this section of the ToL site
Go to detailed links for the ToL site
Go to quick links
Go to quick search
Go to navigation for this section of the ToL site
Go to detailed links for the ToL site