Ichthyosporea
Mesomycetozoa, Mesomycetozoea
Wyth Marshall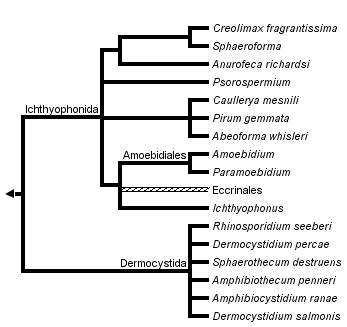


This tree diagram shows the relationships between several groups of organisms.
The root of the current tree connects the organisms featured in this tree to their containing group and the rest of the Tree of Life. The basal branching point in the tree represents the ancestor of the other groups in the tree. This ancestor diversified over time into several descendent subgroups, which are represented as internal nodes and terminal taxa to the right.

You can click on the root to travel down the Tree of Life all the way to the root of all Life, and you can click on the names of descendent subgroups to travel up the Tree of Life all the way to individual species.
For more information on ToL tree formatting, please see Interpreting the Tree or Classification. To learn more about phylogenetic trees, please visit our Phylogenetic Biology pages.
close boxNote: this tree is still under construction. It does not yet contain all known Ichthyosporea subgroups.
Introduction
Ichthyosporeans, equally called mesomycetozoeans, are unicellular osmotrophic feeders. The lineage was first described in 1996 (Ragan et al.) based on four sequenced taxa, and now encompasses over 25 genera (Glocking et al. 2013) (Table 1). All described species have been isolated from or described within an animal host however, sequences are frequently detected in environmental sampling surveys, suggesting either free-living or long lived dispersal stages amongst some clades (del Campo and Ruiz-Trillo 2013). Six genera are in axenic culture (Pirum, Abeoforma, Amoebidium, Ichthyophonus, Sphaeroforma, and Creolimax (Marshall and Berbee 2011, 2013; Marshall et al. 2008; Jostensen et al. 2002; Okamoto et al. 1985; Whisler 1962). Ichthyosporeans are presumed to be the basal most lineage on the branch of the tree of life leading to the multi-cellular animals (Holozoa), and as such, are a valuable group for studying the transition from unicellular to multicellular existence. Recently, following the development of a successful transformation protocol, Creolimax fragrantissima has been proposed as a new model organism (Suga et al. 2013).
Characteristics
The ichthyosporea grow as multinucleate coenocytes and almost always have a cell wall (Figure 1). Depending on the species, environmental condition, stage of life-cycle, or other unknown cues posteriorly flagellate zoospores, spores, or motile amoebae are released (Mendoza et al. 2002) (Table 1). Binary fission and budding have been observed in a few species (Table 1). Motile plasmodia are also found in some cultured species (Okamoto et al. 1985; Marshall et al. 2008; Marshall and Berbee 2011, 2013). Asexual reproduction is predominant but scaliform conjugation, presumably leading to meiosis, was described for Enteropogon sexuale (Hibbits 1978) and genetic evidence of recombination was shown in Sphaeroforma tapetis (formerly Pseudoperkinsus) (Marshall and Berbee 2010). Sphaeroforma was shown to be haploid (Marshall and Berbee 2010), but ploidy has not been described for other species. The composition of the cell walls has not been determined. Carbohydrates are present in Amoebidium parasiticum and Ichthyophonus but neither chitin or cellulose have been identified from wall extracts (Trotter and Whisler, 1965; Franco-Sierra and Alvarez-Pellitero 1999). Other physiology, such as nutritional requirements, are largely unknown.
Ecology and Host Relationships
All described ichthyosporeans were found in association with animals. These relationships range from pathogenic to commensal, and are often not well described. They can either be found embedded within host tissues (e.g. Lohr et al. 2010; Franco-Sierra and Alvarez-Pellitero 1999; Raffel et al. 2008; Pascolini et al. 2003; Savino and Margo 1983), within the digestive lumen (Beebee 1991) or attached to the cuticle of arthropod hindguts (e.g. Mayfield and Lichtwardt 1980; Hibbits 1978). Species described from fish, amphibians, and mammals typically have negative impacts on the host whereas relationships with insect hosts are more likely commensal (Glocking et al. 2013). Population wide effects following epizootics have been described in fish (Kocan et al. 1999). Evidence of emerging pathogenicity has occurred through hitch-hiking on an invasive species (Gozlan et al. 2005) and by pathogen amplification and spillback following the introduction of a naïve host (Hershberger et al. 2010). Broad host ranges have been demonstrated for Ichthyophonus hoferi (Jones and Dawe 2002), Sphaerothecum destruens (Andreou et al. 2012), C. fragrantissima (Marshall et al. 2008), and Sphaeroforma species (Marshall and Berbee 2013). The human parasite, Rhinosporidium seeberi, has also been described in swans, dogs and horses (Kennedy et al. 1995; Leeming et al. 2007; Silva et al. 2005) but ITS variation suggests there may be host correlated strain differences (Silva et al. 2005).
Morphology
In form, ichthyosporeans range from long and thread-like to spherical. Individual cells may reach sizes of 500 micrometres in diameter for some spherical species. Members of some genera (Amphibiothecum, Dermocystidium, Amphibiocystidium, and Rhinosporidium) can form cysts composed of numerous tightly packed spherical cells within the tissues of their hosts (Ashworth 1923; Broz and Privora 1951; Olson et al. 1991; Lotman et al. 2000; Pascolini et al. 2003; Pereira et al. 2005; Raffel et al. 2008). Eccrinales and Amoebidiales secrete holdfasts through pores in their cell walls to attach to their arthropod hosts (Whisler and Fuller 1968; Mayfield and Lichtwardt 1980). The morphology of cultured genera Abeoforma, Pirum, Creolimax and Sphaeroforma is not known under in vivo conditions but Sphaeroforma cells have an external calyx which may function in adhesion (Marshall and Berbee 2013). Amoebae are variously formed with either single prominent pseudopods (C. fragrantissima) or can be more delicately branched as in Abeoforma whisleri. Large motile plasmodia and/or hyphae-like stages have been reported in Dermocystidium koi, Ichthyophonus hoferi, Sphaeroforma tapetis, Creolimax fragrantissima, and Abeoforma whisleri (Dykova and Lom 1992; Franco-Sierra and Alvarez-Pellitero 1999; Marshall et al. 2008; Marshall and Berbee 2013).
Common ultrastructure features include glycogen deposits, membrane bound extensions of the plasma membrane and microtubule organizing centres (Marshall et al 2008; Marshall and Berbee 2011, 2013). Centrioles were observed in Dermocystidium cyprini (Lotman et al. 2000), a species known to produce posteriorly flagellate zoospores. Mitochondria typically have plate-like cristae but tubular cristae are described from Ichthyophonus hoferi (Spanggaard et al. 1996; Franco-Sierra and Alvarez-Pellitero 1999). Cell walls range from thin with single layers, for example in Anurofeca richardsi (Beebee 1991), to multi-layered as in I. hoferi, Psorospermium haeckeli, and A. whisleri (Spanggaard et al 1996; Vogt and Rug, 1999; Marshall and Berbee 2011).
Discussion of Phylogenetic Relationships
The branching order of the unicellular relatives of animals is still in question but most multi-gene phylogenies position the ichthyosporea as the basal-most branch of the metazoan side of the animal-fungus divide (Paps et al. 2013; Toruella et al. 2012; Ruiz-Trillo et al 2006; Steenkamp et al 2006; Ruiz-Trillo et al 2008; Shalchian-Tabrizi et al. 2008). Within the ichthyosporea, there are two clearly distinguished lineages, most commonly referred to as the Ichthyophonida and the Dermocystida. This division is further supported by ecological and morphological differences (Mendoza et al 2002).
Nomenclature
Members of the ichthyosporea were first described in the mid-nineteenth century from arthropod hindguts (e.g. Leidy 1849). New discoveries were misplaced within the fungi, achlorophyllous green algae, or other protist groups until molecular phylogenetic analysis (Baker et al 1999; Benny and O’Donnell 2000; Ustinova 2000; Cafaro 2005). The first recognition of the Ichthyosporea as a group began with the sequencing of four parasites of fish and shellfish (Dermocystidium, the Rosette agent, Ichthyophonus, and Psorospermium), thus inaugurating the lineage with the acronym ‘DRIPs’ (Ragan et al. 1996). In 1998 the clade was given the name ‘Ichthyosporea’ by Cavalier-Smith. Herr et al, (1999), changed this to ‘Mesomycetozoa’ to reflect the phylogenetic affinity near the animal fungus divergence. In 2001, Mendoza et al amended this to ‘Class Mesomycetozoea’. Eukaryote classification schemes still use the original ‘Ichthyosporea’ (Adl et al. 2005, 2012).
Table 1
Table 1. Genera of Ichthyosporea with their hosts and reproductive mechanisms (This information was obtained from the references cited in the text.)
| Genus | Hosts | Reproduction and Dispersal |
| Rhinosporidium | Birds, Mammals | Non-motile spore |
| Dermocystidium | Fish | Zoospore, Non-motile spore |
| Sphaerothecum | Fish | Zoospore, Non-motile spore |
| Anurofeca | Amphibians | Non-motile spore, Binary fission |
| Amphibiothecum | Amphibians | Non-motile spore |
| Amphibiocystidium | Amphibians | Non-motile spore |
| Abeoforma | Mussel | Amoebae, Non-motile spore, Budding, Binary fission |
| Pirum | Sipunculid worm | Non-motile spore |
| Caullerya | Daphnia | Non-motile spore |
| TMS | Beetle | Non-motile spore |
| Creolimax | Various marine invertebrates | Amoebae, Non-motile spore |
| Sphaeroforma | Various marine invertebrates | Non-motile spore |
| Psorospermium | Crayfish | Amoebae, Binary fission |
| Ichthyophonus | Fish | Amoebae, Budding, Non-motile spore |
| Paramobidium | Arthropods | Amoebae, Non-motile spore |
| Amoebidium | Arthropods | Non-motile spore, Amoebae |
| Eccrinidus | Diplopod | Non-motile spore |
| Astreptonema | Amphipod | Non-motile spore |
| Arundinula | Arthropod | Non-motile spore |
| Taeniella | Decapod | Non-motile spore |
| Taeniellopsis | Amphipod | Non-motile spore |
| Enteromyces | Anomura | Non-motile spore |
| Alacrinella | Isopod | Non-motile spore |
| Enterobryus | Arthropod | Non-motile spore |
| Enteropogon | Anomura | Non-motile spore |
| Palavascia | Isopod | Non-motile spore |
References
Adl, S. M., Simpson, A.G.B., Farmer, M.A., Robert A. Andersen, R.A., Anderson, O.R.,. Barta, J.R., Bowser, S.S., et al. 2005. The new higher level classification of eukaryotes with emphasis on the taxonomy of protists. Journal of Eukaryotic Microbiology 52:399-451.
Adl, S.M., Simpson, A.G., Lane, C.E., Luke, J., Bass, D., Bowser, S.S., et al. 2012. The revised classification of eukaryotes. Journal of Eukaryotic Microbiology, 59:429-514.
Andreou, D., Arkush, K.D., Guégan, J-F., Gozlan, R. Introduced pathogens and native freshwater biodiversity: a case study of Sphaerothecum destruens. PLoS ONE 7:e36998.
Ashworth, H. 1923. On Rhinosporidium seeberi (Wernicke, 1903) with special reference to its sporulation and affinities. Transactions of the Royal Society of Edinburgh 53:301-342.
Benny, G.L., O'Donnell, K. 2000. Amoebidium parasiticum is a protozoan, not a Trichomycete. Mycologia, 92:1133-1137.
Baker, G.C., Beebee, T.J., Ragan, M.A. 1999. Prototheca richardsi, a pathogen of anuran larvae, is related to a clade of protistan parasites near the animal-fungal divergence. Microbiology 145:1777-1784.
Beebee, T.J.C. 1991. Purification of an agent causing growth inhibition in anuran larvae and its identification as a unicellular unpigmented alga. Canadian Journal of Zoology 69:2146-2153.
Broz, O., Privora, M. 1951. Two skin parasites of Rana temporaria: Dermocystidium ranae Guyènot Naville and Dermocystidium granulosum n.sp. Parasitology 42:56-69.
del Campo, J., Ruiz-Trillo, I. 2013. Environmental survey meta-analysis reveals hidden diversity among unicellular opisthokonts. Molecular Biology and Evolution 30:802-805.
Cafaro, M.J. 2005. Eccrinales (Trichomycetes) are not fungi, but a clade of protists at the early divergence of animals and fungi. Molecular Phylogenetics and Evolution, 35:21-34.
Cavalier-Smith, T. 1998. Neomonada and the origin of animals and fungi. Pages 375-407 in Coombs, G., Vickerman, K., Sleigh, M., Warren, A. editors. Evolutionary relationships among protozoa. Chapman and Hall, London.
Dykova, I., Lom, J. 1992. New evidence of fungal nature of Dermocystidium koi Hoshina and
Sahara, 1950. Journal of Applied Ichthyology 8,180-185.
Franco-Sierra, A. Alvarez-Pellitero, P. 1999. The morphology of Ichthyophonus sp. in their mugilid hosts (Pisces: Teleostei) following cultivation in vitro. A light and electron microscopy study. Parasitology Research 85, 562-575.
Glocking, S.L., Marshall, W.L., Gleason, F.H. 2013. Phylogenetic interpretations and ecological potentials of the Mesomycetozoea (Ichthyosporea). Fungal ecology 6:237-247.
Gozlan, R.E., St-Hilaire, S., Feist, S.W., Martin, P., Kent, M.L. 2005. Biodiversity: disease threat to European fish. Nature 435:1046.
Herr, R.A., Ajello, L., Taylor, J.W., Arseculeratne, S.N., Mendoza, L. 1999. Phylogenetic analysis of Rhinosporidium seeberi's 18S small subunit ribosomal DNA groups this pathogen among members of the protoctistan Mesomycetozoa clade. Journal of Clinical Microbiology 37:2750-2754.
Hershberger, P.K., van der Leeuw, B.K., Gregg, J.L., Grady, C.A., Lujan, K.M., Gutenberger, S.K., Purcell, M.K., Woodson, J.C., Winton, J.R., Parsley, M.J. 2010. Amplification and transport of an endemic fish disease by an introduced species. Biological invasions 12:3665-3675.
Jones, S.R.M., Dawe, S.C. 2002. Ichthyophonus hoferi Plehn & Mulsow in British Columbia stocks of Pacific herring, Clupia pallasi Valenciennes, and its infectivity to Chinook salmon,Oncorhynchus tshawytscha (Walbaum). Journal of Fish Diseases 25:415-421.
Jostensen, J.P., Sperstad, S., Johansen, S., Landfald, B. 2002. Molecular-phylogenetic, structural and biochemical features of a cold adapted, marine ichthyosporean near the animal-fungal divergence, described from in vitro cultures. European Journal of Protistology 38:93-104.
Kennedy, F.A., Buggage, R.R., Ajello, L. 1995. Rhinosporidiosis: a description of an unprecedented outbreak in captive swans (Cygnus spp) and a proposal for revision of the ontogenic nomenclature of Rhinosporidium seeberi. Journal of Veterinary Mycology 33:157-165.
Kocan, R., Hershberger, P., Mehl, T., Elder, N., Bradley, M., Wildermuth, D., Stick, K. 1999. Pathogenicity of Ichthyophonus hoferi for laboratory-reared Pacific herring Clupea pallasi and its early appearance in wild Puget Sound herring. Diseases of Aquatic Organisms 35:23-29.
Leeming, G., Smith, K.C., Bestbier, M.E., Barrelet, A., Kipar, A. 2007. Equine Rhinosporidiosis in United Kingdom. Emerging Infectioud Diseases 13:1377-1379.
Lohr, J.N., Laforsch, C., Koerner, H., Wolinska, J.A. 2010. Daphnia parasite (Caullerya mesnili) constitutes a new member of the Ichthyosporea, a group of protists near the animal-fungi divergence. Journal of Eukaryotic Microbiology 57:328-336.
Leidy, J. 1849 Enterobryus, a new genus of confervaceae. Proceedings of the National Academy of Sciences Philadelphia 4:225-233.
Lord, J.C., Hartzer, K.L. Kambhampati, S. 2012. A nuptually transmitted Ichthyosporean symbiont of Tenebrio molitor (Coleoptera: Tenebrionidae). Journal of Eukaryotic Microbiology 59:246-250.
Lotman, K., Pekkarinen, M., Kasesalu, J. 2000. Morphological observations on the life cycle of Dermocystidium cyprini Červinka and Lom, 1974, parasitic in carps (Cyprinus carpio). Acta Protozoologica 39:125-134.
Hibbits J. 1978. Marine Eccrinales (Trichomycetes) found in crustaceans of the San Juan Archipelago, Washington. Syesis 11:213-261.
Marshall, W.L., Berbee, M.L. 2010. Population level analyses indirectly reveal cryptic sex and life-history traits of Pseudoperkinsus tapetis (Ichthyosporea, Opisthokonta) a unicellular relative of the animals. Molecular Biology and Evolution 27:2014-2026.
Marshall, W.L., Berbee, M.L. 2011. Facing unknowns: living cultures (Pirum gemmata gen.nov., sp. nov., and Abeoforma whisleri, gen. nov., sp. Nov.) from invertebrate digestive tracts represent an undescribed clade within the unicellular Opisthokont lineage Ichthyosporea (mesomycetozoea). Protist 162:33-57.
Marshall, W.L., Berbee, M.L. 2013. Comparative morphology and genealogical delimitation of cryptic species from sympatric isolates of Sphaeroforma (Ichthyosporea, Opisthokonta). Protist 164:287-311.
Marshall, W.L., Celio, G., McLaughlin, D.J., Berbee, M.L. 2008. Multiple isolations of a culturable, motile Ichthyosporean (Mesomycetozoa, Opisthokonta), Creolimax fragrantissima, n. gen., n. sp., from marine invertebrate digestive tracts. Protist 159:415-433.
Mayfield, S.D., Lichtwardt, R.W. 1980. Comparative study of the holdfast structure in four Trichomycetes. Canadian Journal of Botany 58:1074-1087.
Mendoza, L., Ajello, L., Taylor, J. 2001. The taxonomic status of Lacazia loboi and Rhinosporidium seeberi has finally been resolved with the use of molecular tools. Revista Iberoamericana de Micologia 18:95-98.
Mendoza, L., Taylor, J.W., Ajello, L. 2002. The class Mesomycetozoea: a heterogeneous group of microorganisms at the animal-fungal boundary. Annual Reviews in Microbiology 56:315-344.
Okamoto, N., Nakase, K., Suzuki, H., Nakai, Y., Fujii, K., Sano, T. 1985. Life history and morphology of Ichthyophonus hoferi in vitro. Fish Pathology 20:273-285.
Olson, R.E., Dungan, C.F., Holt, R.A. 1991. Water-borne transmission of Dermocystidium salmonis in the laboratory. Diseases of Aquatic Organisms 12:41-48.
Ordas, M.C., Novoa, B., Faisal, M., McLaughlin, S., Figueras, A. 2001. Proteolytic activity of cultured Pseudoperkinsus tapetis extracellular products. Comparative Biochemistry and Physiology Part B 130:199-206.
Raffel, T.R., Bommarito, T., Barry. D.S., Witiak, S.M., Shackelton, L.A. 2008. Widespread infection of the Eastern red-spotted newt (Notophthalnus viridescens) by a new species of Amphibiocystidium, a genus of fungus-like mesomycetozoan parasites not previously reported in North America. Parasitology 135:203-215.
Ragan, M.A., Goggin, C.L., Cawthorn, R.J., Cerenius, L., Jamieson, A.V., Plourde, S.M., Rand, T.G., Soderhall, K., Gutell, R.R. 1996. A novel clade of protistan parasites near the animal-fungal divergence. Proceedings of the National Academy of Sciences, 93:11907-11912.
Pascolini, R., Daszak, P., Cunningham, A.A., Tei, S., Vagnetti, D., Bucci, S., Fagotti, A., Di Rosa, I. 2003. Parasitism by Dermocytidium ranae in a population of Rana esculenta complex in Central Italy and description of Amphibiocystidium n. gen. Diseases of Aquatic Organisms 56:65-74.
Pereira, C.N., Di Rosa, I., Fagotti, A., Simoncelli, F., Pascolini, R., Mendoza, L. 2005. The pathogen of frogs Amphibiocystidium ranae is a member of the Order Dermocystida in the Class Mesomycetozoea. Journal of Clinical Microbiology 43: 192-198.
Ruiz-Trillo, I., Lane, C.E., Archibald, J M., Roger, A.J. 2006. Insights into the evolutionary origin and genome architecture of the unicellular opisthokonts Capsaspora owczarzaki and Sphaeroforma arctica. Journal of Eukaryotic Microbiology 53:379-384.
Ruiz-Trillo, I., Roger, A.J., Burger, G., Gray, M.W., Lang, B. F. 2008. A phylogenomic investigation into the origin of metazoa. Molecular Biology and Evolution 25:664-672.
Shalchian-Tabrizi, K., Minge, M.A., Espelund, M., Orr, R., Ruden, T., Jakobsen, K.S., Cavalier-Smith, T. 2008. Multigene phylogeny of choanozoa and the origin of animals. PloS one, 3:e2098.
Silva, V., Pereira, C.N., Ajello, L., Mendoza, L. 2005. Molecular evidence for multiple host-specific strains in the genus Rhinosporidium. Journal of Clinical Microbiology 43:1865-1868.
Steenkamp, E.T., Wright, J., Baldauf, S.L. 2006. The protistan origins of animals and fungi. Molecular Biology and Evolution, 23:93-106.
Torruella, G., Derelle, R., Paps, J., Lang, B.F., Roger, A.J., Shalchian-Tabrizi, K., Ruiz-Trillo, I. 2012. Phylogenetic relationships within the Opisthokonta based on phylogenomic analyses of conserved single-copy protein domains. Molecular Biology and Evolution, 29:531-544.
Trotter, M.J., Whisker, H.C. 1965. Chemical composition of the cell wall of Amoebidium parasiticum. Canadian Journal of Botany 43:869-876.
Paps, J., Medina-Chacon, L.A., Marshall, W.L., Suga, H., Ruiz-Trillo, I. 2013, Molecular phylogeny of Unikonts: new insights into the position Apusozoa and the internal relationships of opisthokonts. Protist 164:2-12.
Ragan, M.A., Goggin, C.L., Cawthorn, R.J., Cerenuis, L., Jamieson, A.V.C., Plourde, S.M., Rand, T.G., Soderhall, K., Gutell, R.R. 1996. A novel clade of protistan parasites near the animal-fungal divergence. Proceedings of the National Acadamy of Sciences USA 93:11907-11912.
Savino, D.F., Margo, C.E. 1983. Conjunctival Rhinosporidiosis: light and electron microscopic study. Ophthalmology 90:1482-1489.
Spanggaard, B., Skouboe, P., Rossen, L., Taylor, J.W. 1996. Phylogenetic relationship of the intracellular fish pathogen Ichthyophonus hoferi, and fungi, choanoflagellates and the rosette agent. Marine Biology 126: 109-115.
Suga, H., Ruiz-Trillo, I. 2013. Development of icthyosporeans sheds light on the origin of metazoan multicellularity. Developmental Biology doi:10.1016/j.ydbio.2013.01.009.
Ustinova, I., Krienitz, L., Huss, V.A.R.. 2000. Hyaloraphidium curvatum is not a green alga, but a lower fungus; Amoebidium parasiticum is not a fungus, but a member of the DRIPs. Protist 151:253-262.
Vogt, G., Rug, M. 1999. Life stages and tentative life cycle of Psorospermium haeckeli, a species of the novel DRIPs clade from the animal-fungal dichotomy. Journal of experimental Zoology 283:31-42.
Whisler, H.C. 1962. Culture and nutrition of Amoebidium parasiticum. American Journal of Botany 49:193-199.
Whisler, H.C., Fuller, M.S. 1968. Preliminary observations on the holdfast of Amoebidium parasiticum. Mycologia 60:1068-1079.
About This Page

University of British Columbia, Vancouver, British Columbia, Canada
Correspondence regarding this page should be directed to Wyth Marshall at
Page copyright © 2014
 Page: Tree of Life
Ichthyosporea. Mesomycetozoa, Mesomycetozoea.
Authored by
Wyth Marshall.
The TEXT of this page is licensed under the
Creative Commons Attribution-NonCommercial License - Version 3.0. Note that images and other media
featured on this page are each governed by their own license, and they may or may not be available
for reuse. Click on an image or a media link to access the media data window, which provides the
relevant licensing information. For the general terms and conditions of ToL material reuse and
redistribution, please see the Tree of Life Copyright
Policies.
Page: Tree of Life
Ichthyosporea. Mesomycetozoa, Mesomycetozoea.
Authored by
Wyth Marshall.
The TEXT of this page is licensed under the
Creative Commons Attribution-NonCommercial License - Version 3.0. Note that images and other media
featured on this page are each governed by their own license, and they may or may not be available
for reuse. Click on an image or a media link to access the media data window, which provides the
relevant licensing information. For the general terms and conditions of ToL material reuse and
redistribution, please see the Tree of Life Copyright
Policies.
- First online 06 December 2014
- Content changed 06 December 2014
Citing this page:
Marshall, Wyth. 2014. Ichthyosporea. Mesomycetozoa, Mesomycetozoea. Version 06 December 2014 (under construction). http://tolweb.org/Ichthyosporea/121172/2014.12.06 in The Tree of Life Web Project, http://tolweb.org/





 Go to quick links
Go to quick search
Go to navigation for this section of the ToL site
Go to detailed links for the ToL site
Go to quick links
Go to quick search
Go to navigation for this section of the ToL site
Go to detailed links for the ToL site