Dicamptodontidae
Pacific Giant Salamanders
Allan Larson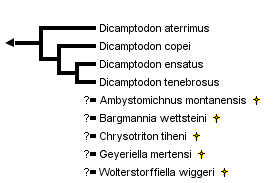


This tree diagram shows the relationships between several groups of organisms.
The root of the current tree connects the organisms featured in this tree to their containing group and the rest of the Tree of Life. The basal branching point in the tree represents the ancestor of the other groups in the tree. This ancestor diversified over time into several descendent subgroups, which are represented as internal nodes and terminal taxa to the right.

You can click on the root to travel down the Tree of Life all the way to the root of all Life, and you can click on the names of descendent subgroups to travel up the Tree of Life all the way to individual species.
For more information on ToL tree formatting, please see Interpreting the Tree or Classification. To learn more about phylogenetic trees, please visit our Phylogenetic Biology pages.
close boxRelationships among the four extant species of the family Dicamptodontidae are based upon the allozymic study of Good (1989). For a review of the extinct species, see Estes (1981).
Introduction
The salamander family Dicamptodontidae contains four extant species placed in the single genus, Dicamptodon (Good, 1989) plus extinct species placed in the genera Ambystomichnus, Bargmannia, Chrysotriton, Geyeriella and Wolterstorffiella (Estes, 1981). Dicamptodons are commonly known as Pacific giant salamanders because they are found along the Pacific coast of western North America, and they attain relatively large sizes (body length approaching 6 inches excluding the tail). They inhabit coniferous forests of northwestern California, Oregon, Washington, Idaho and British Columbia (Good, 1989). Larvae inhabit springs and streams, where they can be found in pools under rocks. Metamorphosed adults are found under logs, bark or stones or crawling exposed on the forest floor. Adults are active particularly on wet nights following a strong rain. Members of one species, Dicamptodon copei, lack metamorphosis and retain gills and an aquatic habitat throughout life.
Fossil remains indicate that the Dicamptodontidae formerly had a broader geographic range than indicated by extant species (see Estes, 1981). Fossils extend farther east in North America (Montana and North Dakota) than extant species and are reported also from Europe (Germany and Slovakia).
Dicamptodon and its fossil relatives formerly were placed in the salamander family Ambystomatidae and later moved with Rhyacotriton to the family Dicamptodontidae. Molecular phylogenetic studies (Larson, 1991; Larson and Dimmick, 1993) showed that Dicamptodon is a close relative of Ambystoma and conceivably could have been left in the family Ambystomatidae, but that Rhyacotriton is only a distant relative of both genera and should not be grouped with them at the family level. Rhyacotriton is now placed in the separate family Rhyacotritonidae (Good and Wake, 1992).
Characteristics
Diagnosis
Dicamptodontid salamanders resemble ambystomatids but attain larger adult sizes (up to about 6 inches in body length excluding the tail). Larvae have short external gills and 4 pairs of gill slits, and a dorsal finlike projection on the tail. Some forms retain the larval appearance into adulthood; others metamorphose into terrestrial adults that have robust bodies and limbs, broad heads with prominent eyes, and a laterally-flattened tail. The dorsal surface of the body usually has a mottled brown and black pattern.
Detailed Characteristics of the Dicamptodontidae
The morphological characters given below are the ones standardly used to diagnose the salamander family Dicamptodontidae and to assess its phylogenetic relationships to other salamanders. The individual characteristics are in most cases shared with other salamanders and should not be interpreted as synapomorphies of the Dicamptodontidae. Absence of characteristics found in other salamanders is noted where it is important for distinguishing dicamptodontids from other salamanders and/or determining their relationships to other salamanders. These characteristics were assembled from a large number of original sources by Duellman and Trueb (1986), Larson (1991) and Larson and Dimmick (1993).
Morphology of the Skull
The premaxilla consists of separated, paired bones. Bilaterally paired nasal bones each ossify from a single, laterally positioned anlage; long posterior processes of the premaxillae extend between the paired nasal bones and completely separate them. Maxillary bones are present and well developed. Bilaterally paired septomaxillary bones are present. Lacrimal bone is present. Quadratojugal bone is absent. Pterygoid bones are present. An internal carotid foramen is present in the parasphenoid bone. The angular bone is fused to the mandible. The columella and operculum appear as separate ear bones detached from the otic capsule. Replacement of vomerine teeth proceeds from the posterior of the vomer. Teeth have a distinct crown and pedicel. Origin of the levator mandibulae anterior superficialis muscle includes the exoccipital.
Inner Ear
A basilaris complex is present in the inner ear. The recessus amphibiorum is oriented horizontally in the inner ear. The otic sac is bulbar and unvascularized. The amphibian periotic canal lacks fibrous connective tissue. The periotic cistern is large and protrudes into the fenestra.
Hyobranchial structures
The first hypobranchial and first ceratobranchial (alternatively homologized as the first ceratobranchial and first epibranchial, respectively) exist as separate structures. The second ceratobranchial (alternatively homologized as the second epibranchial) comprises a single element. The otoglossal cartilage is present as a plate in the hyoid apparatus of adults. Lungs and an ypsiloid cartilage are present. Larvae have four pairs of gill slits.
Characteristics of the Trunk and Vertebral Column
The scapula and coracoid bones of the pectoral girdle are fused to form the scapulocoracoid. Vertebral centra are amphicoelous. Ribs are bicapitate. Only the sacral and postsacral vertebrae have spinal-nerve foramina in their neural arches (foramen is partial in sacral vertebra). Anterior glomeruli of the kidney are reduced or absent.
Reproductive characters
Fertilization is internal. Ciliated epithelium is present in the cloacal tube and/or anterior cloacal chamber of females. Epidermal lining is absent from the anterior cloacal chamber of females. Evaginations are present in the dorsolateral walls of the male cloacal tube. Anterior ventral glands are present in the cloacae of females. Spermathecae are present in the female cloacal chamber. Glands secreting into the dorsal walls of the female cloaca are absent. Anterior ventral glands are present in male cloacae. Posterior ventral glands are present in male cloacae. Kingsbury's glands are present in male cloacae. Dorsal pelvic glands are present in males. Lateral pelvic glands are present in males. Glands secreting into the male cloacal orifice are present. Parental care of eggs is by females.
The diploid number of chromosomes is 28.
Classification
The family Dicamptodontidae is part of the salamander suborder Salamandroidea, which contains all of the internally fertilizing salamanders (Duellman and Trueb, 1986). The family Rhyacotritonidae formerly was recognized as the subfamily Rhyacotritoninae of the family Dicamptodontidae, but was given family-level status by Good and Wake (1992) following evidence that the Dicamptodontidae as formerly recognized was not monophyletic (Larson, 1991; Larson and Dimmick, 1993). The family Dicamptodontidae as currently recognized contains what formerly was considered the subfamily Dicamptodontinae (Dicamptodon plus the extinct genera Ambystomichnus, Bargmannia, Chrysotriton, Geyeriella and Wolterstorffiella) prior to removal of Rhyacotriton to a separate family.
Discussion of Phylogenetic Relationships
Knowledge of phylogenetic relationships among the four extant species of Dicamptodon comes from the allozymic studies of Daugherty et al. (1983) and Good (1989). The phylogenetic relationships shown are as revealed Good's (1989) parsimony analysis of allozymic data using presence versus absence of alleles as characters. Although neither allozymic study used an outgroup to root the tree, the genetic distance separating the Idaho populations (D. aterrimus) from the coastal ones (D. copei, D. ensatus and D. tenebrosus) is much higher than genetic distances among populations within either of these geographical regions; therefore, the earliest phylogenetic split among lineages leading to extant Dicamptodon appears to have separated D. aterrimus from the other species. The exact placement of the three coastal species relative to each other is more tentative; alternative methods of phylogenetic analysis of the same data yield conflicting groupings of species (Good, 1989).
References
Daugherty, C. H., F. W. Allenndorf, W. W. Dunlap and K. L. Knudsen. 1983. Systematic implications of geographic patterns of genetic variation in the genus Dicamptodon. Copeia 1983:679-691.
Duellman, W. E. and L. Trueb. 1986. Biology of Amphibians. McGraw-Hill, New York.
Estes, R. 1981. Gymnophiona, Caudata. Handbuch der Paläoherpetologie 2:1-115.
Good, D. A. 1989. Hybridization and cryptic species in Dicamptodon (Caudata: Dicamptodontidae). Evolution 43:728-744.
Good, D. A. and D. B. Wake. 1992. Geographic variation and speciation in the torrent salamanders of the genus Rhyacotriton (Caudata: Rhyacotritonidae). University of California Publications in Zoology 126:1-91.
Larson, A. 1991. A molecular perspective on the evolutionary relationships of the salamander families. Evolutionary Biology 25:211-277.
Larson, A. and W. W. Dimmick. 1993. Phylogenetic relationships of the salamander families: A analysis of congruence among morphological and molecular characters. Herpetological Monographs 7:77-93.
Title Illustrations

| Scientific Name | Dicamptodontidae |
|---|---|
| Location | Mendocino County, California |
| Comments | The Pacific Giant Salamander |
| Specimen Condition | Live Specimen |
| Image Use |
 This media file is licensed under the Creative Commons Attribution-NonCommercial-ShareAlike License - Version 3.0. This media file is licensed under the Creative Commons Attribution-NonCommercial-ShareAlike License - Version 3.0.
|
| Copyright |
© 1996 Kevin de Queiroz

|
About This Page
Kevin de Queiroz, David Heyse, Tomas Hrbek and Todd Jackman contributed to the preparation of this Tree of Life page.
Allan Larson

Washington University, St. Louis, Missouri, USA
Page copyright © 1996 Allan Larson
All Rights Reserved.
Citing this page:
Larson, Allan. 1996. Dicamptodontidae. Pacific Giant Salamanders. Version 01 January 1996 (under construction). http://tolweb.org/Dicamptodontidae/15447/1996.01.01 in The Tree of Life Web Project, http://tolweb.org/







 Go to quick links
Go to quick search
Go to navigation for this section of the ToL site
Go to detailed links for the ToL site
Go to quick links
Go to quick search
Go to navigation for this section of the ToL site
Go to detailed links for the ToL site