Architeuthidae , Architeuthis
Architeuthis dux
Clyde F. E. RoperTwenty nominal species have been described in the family Architeuthididae (Clarke, 1966), but the systematics of the group is so poorly known that the actual number of valid species has long been uncertain. A morphological study (Försch, 1998), however, found no convincing evidence for more than one species. A recent molecular analysis (Winkelmann, et al., 2013) reached the same conclusion that probably only one species is valid.
- Architeuthis dux
- Architeuthis martensi
- Architeuthis sanctipauli
Introduction
Architeuthis dux is the largest (in terms of weight) known cephalopod, the largest known mollusks and probably the largest invertebrates ever known to exist in the oceans. Largest specimens attain mantle lengths up to 5 m and total lengths up to 18 m, but animals of that size are seldom reported. Most records are in the 6-12 m total length range. A. dux is characterized by having small, ovoid fins, very long arms, exceptionally long tentacles, and distinctive tentacular club structure. The only squid that approaches A. dux in size is the cranchiid Mesonychoteuthis hamiltoni.
Reviews are available of giant squid morphology, biology and distribution (Roper & Boss, 1982), occurrence and morphology in southern African waters (Roeleveld & Lipinski, 1991), systematics and biology in Newfoundland (Aldrich, 1992) and New Zealand (Försch, 1998) with a re-evaluation of species world-wide. The huge size of these animals and their (former) rarity, have engendered numerous myths and mysteries, both concerning their dimensions and their antagonistic, violent behavior towards ships, sailors and fishermen. Some of these accounts go back to the earliest natural history volumes published in the mid-1500s. The books by Lane (1960) and Ellis (1995, 1998) chronicle many of these stories.
Diagnosis
An oegopsid ...
- with buccal connectives attaching to dorsal borders of arms IV.
- with dense cluster of small suckers in 6-7 irregular series on carpal region of club with interspersed locking knobs.
Characteristics
- Arms
- Arms with suckers in two series.
- Buccal connectives attach to dorsal borders of arms IV.
- Tentacles
- Tentacular clubs elongate, narrow, with suckers in four longitudinal series.
- Tentacular clubs divided into distinct carpus, manus and dactylus.
- Manus with enlarged suckers in medial two rows.
- Carpal region with dense cluster of suckers, in 6-7 irregular series; suckers with interspersed hemispherical knobs.
- Alternating pairs of suckers and knobs distributed along nearly entire length of tentacle stalk, becoming more closely-set from proximal to distal ends.
 Click on an image to view larger version & data in a new window
Click on an image to view larger version & data in a new window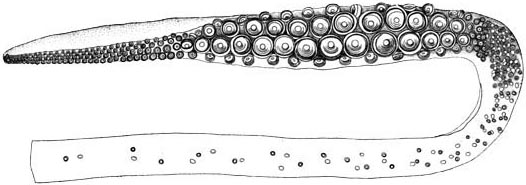
Figure. Oral view of distal region of the tentacle of Architeuthis, Drawing from Verrill (1879).
- Head
- Beaks: Descriptions can be found here: Lower beak; upper beak.
- Beaks: Descriptions can be found here: Lower beak; upper beak.
- Fins
- Photophores
- Photophores absent.
Nomenclature
Many, if not most, species of Architeuthis have been described from a single specimen that was discovered stranded on shore, floating on the surface, or taken from the stomach of a sperm whale. Until the 1980s, captures of specimens in fishing nets were very rare. Consequently, no type specimen has been complete, and some species have been named solely on the basis of parts only, e.g., a mangled head, or a single beak. Without comparable characters, and with so badly damaged specimens, early workers had no basis for comparison with other “species”, so they would name yet another new species of Architeuthis.
Not only were many species named in the architeuthids, but eight different genera were created, as well. These were:
- Architeuthis Steenstrup, 1857
- Megaloteuthis Kent, 1874
- Dinoteuthis More,1875
- Mouchezis Velain, 1877
- Megateuthis Hilgendorf,1880
- Plectoteuthis Owen, 1881
- Steenstrupia Kirk,1882
- Dubioteuthis Joubin,1899
All these genera are synonyms of Architeuthis Steenstrup, 1857.
Nesis, (1982/1987) and others consided that perhaps three valid species exist. But they are not well defined nor differentiated. These are A. dux Steenstrup, 1857 in the North Atlantic Ocean, A. martensi (Hilgendorf, 1880) in the North Pacific and A. sanctipauli (Verlain, 1877) in the Southern Ocean. The more recent data of Forsch (1998) and Winkelmann et al. (2013) indicate that only one species with a world-wide distribution exists.
A list of all nominal genera and species in the Architeuthidae can be found here. The list includes the current status and type species of all genera, and the current status, type repository and type locality of all species and all pertinent references.
Life History
The early life history of Architeuthis is virtually unknown, beginning with mating, fertilization, egg laying, embryonic development, hatching, larval development. Are the eggs laid in huge gelatinous masses or individually? Only one paralarva has been reported to date: Lu (1986) recorded a single specimen of 10.3 mm ML taken in the Tasman Sea off Australia at a depth of 20 m but provided no definitive characteristics linking the specimen to Architeuthis. The condition of the specimen did not permit a detailed illustration or description, so it is difficult to derive conclusions about this form.
Two juveniles of Architeuthis were described from specimens of 57 mm ML from off Madeira Island, eastern North Atlantic and 45mm ML from the eastern Pacific Ocean off Chile (Roper and Young, 1972). Both were taken from the stomachs of deep-sea fishes, Alepisaurus ferox, and exhibited marked differences in body proportions suggesting that they may represent two different species. The Atlantic specimen has arms as long as the mantle, while in the Pacific form the longest arms (II-IV) are about 60% of the ML; the Pacific specimen has tentacles less than half as long as the Atlantic form.. These specimens when reported were an order of magnitude smaller than the previously known smallest architeuthid.

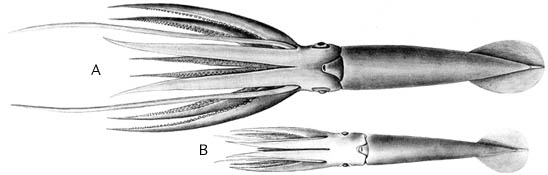
Figure. Oral view of two juvenile Architeuthis. A - Atlantic specimen. B - Pacific specimen. Drawings from Roper and Young (1972).
Several small specimens have been reported as architeuthids, but close inspection reveals them to belong to other families. One specimen reported by Toll & Hess (1981) as Architeuthis is a mature male at 167 mm ML, a remarkably small size for a true architeuthid.
Cephalopods are known to be very fast growing animals. Some species of small, shallow water forms reach sexual maturity in 6-8 months, and most species about which growth, age and maturity data are available reach reproductive capacity within 12-18 months. Many of the specimens of Architeuthis that have been recovered have been mature, especially the females (which in any case are much more abundant in collections). But the age at maturity of Architeuthis is not known with certainty. One study suggests that adult size is attained within 3 years (Gauldie, et al., 1994). Even at the rapid growth rate expected in cephalopods, the attainment of a mass of 500 kg or more in fewer than 3 years is impressive.
Females produce enormous quantities of whitish to cream-colored eggs, about 0.5-1.4 mm long and 0.3-0.7 mm wide, depending on the stage of their maturity. One female had over 5000 gm (over 11 pounds) of eggs in her ovary, well in excess of a million eggs. As in most oegopsids, females have a single median ovary in the posterior end of the mantle cavity, paired, convoluted oviducts along which mature eggs pass, then exit through the oviducal glands, and large nidamental glands that produce quantities of gelatinous material. Whether the eggs are laid into a large gelatinous matrix, as in most of the large oceanic squids, e.g., ommastrephids and thysanoteuthids, or are released individually, is unknown, although the large nidamental glands suggest the former method (Roper & Boss, 1982 and new information).
Males tend to reach sexual maturity at a smaller size than do females. The two ventral arms, arms IV, are reported to be modified (=hectocotylized) to transfer very long, thin, cylindrical packets of sperm, the spermatophores, to the female. As in most other cephalopods, the single, posterior testis produces sperm that move into a complex system of glands that manufacture the spermatophores. These are stored in an elongate sac, Needham’s sac, from which they are expelled during mating. The Needham’s sac of fully mature males is packed with hundreds of spermatophores. Needham’s sac terminates in the penis. The penis is so elongate that it extends anteriorly beyond the mantle opening. While mating has not been observed and the exact role of the penis is uncertain, some females have been found with spermatangia, the sperm-containing sacs of the spermatophore, embedded in the tissue around the bases of the arms and the head (Norman & Lu, 1997).
Since the advent of commercial deep-sea trawling for orange roughy, hoki and scampi in New Zealand and Australian waters in the early 1980s, a number of Architeuthis specimens have been captured and landed for teuthologists to examine. Total lengths range from 3-4 m to a maximum of 13-14 m. In many cases, not only from trawls but from the other sources, the smaller specimens tend to be males. The reason for this must await further analysis and sufficient material from a single geographic region.
Distribution
Vertical distribution
Since most records of Architeuthis prior to the late 20th century are from strandings, floaters and sperm whale stomachs, it is difficult to assess the vertical distribution of the architeuthids. Indirect evidence has suggested a deep-sea habitat, and deep net captures, as well as knowledge of sperm whale foraging behavior, now confirm that the depth distribution of these huge animals must occupy a considerable range. Certainly their occurrence in the zone of 300 to 1000 m is not unrealistic. And captures in both bottom trawls and in midwater trawls far off the bottom indicate a broad range of habitat selection.
Geographical distribution
The Architeuthidae are now known to be distributed around the world, usually in association with continental and island slopes. Concentrations of records are from the North Atlantic Ocean, especially Newfoundland, Norway, northern British Isles and the oceanic islands of the Azores and Madeira; the South Atlantic in southern African waters; the North Pacific around Japan, and the southwestern Pacific around New Zealand and Australia; circumglobal in the Southern Ocean. Specimens are rare from tropical and high polar latitudes.
References
Aldrich, F. A. 1992. Some aspects of the systematics and biology of squid of the genus Architeuthis based on a study of specimens from Newfoundland waters. Bulletin of Marine Science, 49 (1-2): 457-481.
Clarke, M.R. 1966. A review of the systematics and ecology of oceanic squids. Advances in Marine Biology, 4: 91-300.
Ellis, R. 1995. Monsters of the Sea. Knopf
Ellis, R. 1998. The search for the giant squid. The Lyons Press, NY, 322 pp.
Försch, E. C. 1998. The marine fauna of New Zealand: Cephalopoda: Oegopsida: Architeuthidae (giant squid). NIWA Biodiversity Memoir 110. 113 pp.
Gauldie, R.W., I.F. West and E.C. Forch, 1994. Statocyst, statolith, and age estimation of the giant squid, Architeuthis kirki. The Veliger, 37(1):93-109.
Lane, F. W. 1960. Kingdom of the Octopus. The Life History of Cephalopoda. Jarrolds, New York. 300pp.
Lindgren, A. R. 2010. Molecular inference of phylogenetic relationships among Decapodiformes (Mollusca: Cephalopoda) with special focus on the squid Order Oegopsida. Mol. Phylogenet. Evol. (2010), 56 (1): 77-90.
Lindgren, A. R., M. S. Pankey, F. G. Hochberg and T. H. Oakley. 2012. A multi-gene phylogeny of Cephalopoda supports convergent morphological evolution in association with multiple habitat shifts in the marine environment. BMC Evolutionary Biology 2012, 12:129-144.
Lu, C.C. 1986. Smallest of the largest—first record of giant squid larval specimen. Australian Shell News, 53:9.
Nesis, K.N. 1982/1987. Abridged key to the cephalopod mollusks of the world’s oceans. Light and Food Industry Publishing House, Moscow, 358pp. [In Russian]. Cephalopods of the World; Squids, Cuttlefishes, Octopuses, and Allies. T.F.H. Publications, Inc., Neptune City, NJ, USA, 351 pp.
Roeleveld, M.C. & M.R. Lipinski, 1991. The giant squid Architeuthis in southern African waters. Journal Zoology London, 224: 431-477.
Roper, C.F.E. & R.E. Young, 1972. First records of juvenile giant squid, Architeuthis (Cephalopoda: Oegopsida). Proceedings of the Biological Society of Washington, 85(16): 205-222.
Roper, C.F.E. & K.J. Boss, The giant squid. Scientific American, 246(4): 96-105.
Toll, R.B. & S.C. Hess, A small, mature male Architeuthis (Cephalopoda: Oegopsida), with remarks on maturation in the family. Proceedings of the Biological Society of Washington, 94: 753-760.
Verrill, A. E. 1879. The cephalopods of the north-eastern coast of America. Part I. The gigantic squids (Architeuthis) and their allies; with observations on similar large species from foreign localities. Trans. Connecticut Acad. Sciences, 5: 23: 177-257.
Winkelmann, I., F. Campos, J. Strugnel, Y. Cherel, P. Smith, T. Kubodera, L. Allcock, M. Kampmann, H. Schroeder, A. Guerra, M. Norman, J. Finn, D. Ingrao, M. Clarke and M. gilbert. 2013. Mitochondrial genome diversity and population structure of the giant squid Architeuthis: genetics sheds new light on one of the most enigmatic marine species. Proc. Royal Society B, 280. http://dx.doi.org/10.1098/rspb.2013.0273
Information on the Internet
- Search for Giant Squid. An Expedition into the Last Frontier. About the 1999 Smithsonian expedition to New Zealand.
- In Search of Giant Squid. Another web site about Smithsonian giant squid expeditions.
- Architeuthis (Giant Squid) reproduction, with notes on basic anatomy and behavior. TONMO.com article by Steve O'Shea
- It Came From Down Under. Giant squid at the American Museum of Natural History.
Title Illustrations

| Scientific Name | Architeuthis |
|---|---|
| Location | New Zealand |
| Comments | Steve O'Shea holding the tentacle of an Architeuthis sp. |
| Copyright | © 1996 Martin Hunter, New Zealand Herald |
| Scientific Name | Architeuthis |
|---|---|
| Location | off south Australia |
| Sex | Female |
| Size | 12m long (total length) |
| Copyright |
© 1996

|
| Scientific Name | Architeuthis "princeps" |
|---|---|
| Comments | Modified from Verrill, 1879. |
| Reference | Verrill, A. E. 1879. The cephalopods of the north-eastern coast of America. Part I. The gigantic squids (Architeuthis) and their allies; with observations on similar large species from foreign localities. Trans. Connecticut Acad. Sciences 5:23:177-257. |
About This Page

Smithsonian Institution, Washington, D. C., USA
Page copyright © 2019
All Rights Reserved.
- Content changed 27 February 2016
Citing this page:
Roper, Clyde F. E. 2016. Architeuthidae , Architeuthis . Architeuthis dux . Version 27 February 2016. http://tolweb.org/Architeuthis_dux/19408/2016.02.27 in The Tree of Life Web Project, http://tolweb.org/













 Go to quick links
Go to quick search
Go to navigation for this section of the ToL site
Go to detailed links for the ToL site
Go to quick links
Go to quick search
Go to navigation for this section of the ToL site
Go to detailed links for the ToL site